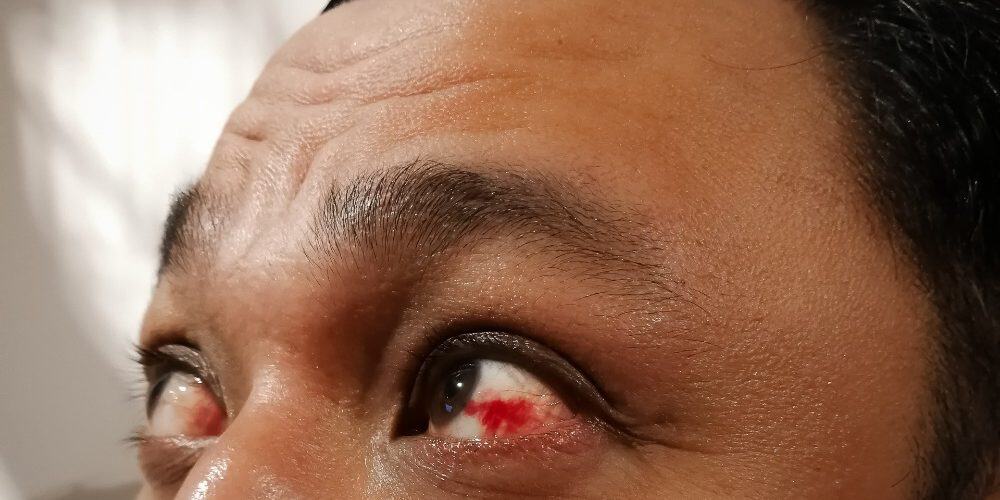
Faricimab Under Review for Macular Edema Following Retinal Vein Occlusion
The Food and Drug Administration (FDA) has accepted the supplemental Biologics License Application (sBLA) for faricimab-svoa for the treatment of macular edema following retinal vein occlusion (RVO).
The sBLA is supported by data from the randomized, multicenter, double-masked phase 3 BALATON (ClinicalTrials.gov Identifier: NCT04740905) and COMINO (ClinicalTrials.gov Identifier: NCT04740931) studies, which included 553 patients with macular edema due to branch RVO and 729 patients with macular edema secondary to central retinal or hemiretinal vein occlusion, respectively. Patients were randomly assigned 1:1 to receive 6 monthly injections of either faricimab 6mg or aflibercept 2mg for 20 weeks; from weeks 24 to 72, all patients received faricimab 6mg up to every 4 months.




![[column] Toekomst tijdschriften glanst als nooit tevoren](https://www.pharmamarketeer.nl/wp-content/uploads/2024/12/happy-couple-in-t-shirts-reading-newspaper-while-h-2024-11-10-00-13-23-utc-1-80x60.jpg)
![[column] MarketingAffairs 17: Marketing als oplossing](https://www.pharmamarketeer.nl/wp-content/uploads/2024/12/asian-seo-manager-writing-on-glass-with-illustrati-2024-11-17-23-11-51-utc-1-80x60.jpg)


